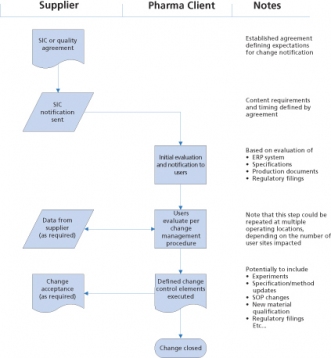
To ensure that requirements for reporting of changes are understood and agreed to, pharmaceutical and biotech manufacturers will typically require suppliers to enter into an agreement, sometimes referred to as a supplier-initiated change or SIC agreement, as a pre-requisite for doing business. In some cases, it may take the form of a quality agreement, which is discussed later. An example generic SIC agreement is included in Figure 1. This agreement establishes the need for and timing of reporting. The types of changes that are subject to be reported are specific to the material or services purchased, and include but are not limited to the following:
All suppliers
• Change in address or location (manufacturing, testing, and warehousing among others)
• Change in ownership
• Change in company name
• Use of a new subcontractor for purchased materials or services
• Change in certification or accreditation status by standard setting body or regulatory agency (e.g., International Organization for Standardization [ISO], FDA, EMEA, American National Standards Institute [ANSI]).
Material suppliers (including product-contact disposables)
• Change in raw material supplier or source in packaging configuration or components
• Chnage in packaging confirmation or components
• Change in part number
• Change in product documentation (certificate of analysis, certificate of compliance, product drawing, content on the primary container label)
• Change in raw material supplier or grade (e.g., United States Pharmacopeia [USP], National Formulary [NF], multi-compendial, etc.)
• Change in manufacturing process (e.g., addition/deletion of unit operations, significant change in qualified equipment), including but not limited to:
o Changes in process or chemistry that result in a physical property change
o Changes in purification and/or isolation steps where the result is a less pure product going into the final isolation/purification step
o Changes in manufacturing process that causes a change to the end product impurity profile
• Discontinuation of a material
• Change in specification
• Change in storage condition or shelf life
• Introduction of other processing, such as non-pharmaceutical products, penicillin, hormones, pesticides, or other potent compounds (including phthalates and heavy metals) into multi-use equipment or building
• Changes in sterilization, including “like-for-like” equipment, changes to cycle, criteria, or methods
• Change that results in the potential for cross-contamination a purchased item, including the introduction of a new product or grade of product on equipment that was previously dedicated
• Change to the animal-origin status of a material, change to the country of origin or tissue of an animal-derived material.
Testing Laboratory or Calibration Service Provider
• Failure or out-of-tolerance (OOT) condition of a standard or instrument used for calibration or testing of equipment or material
• Change in test method or documentation
• Re-qualification or re-validation of a test method.
Material Distributor or Warehousing
• Use of a new facility for storage or distribution of materials
• Change in processes or qualified equipment impacting storage and distribution of product
• Failure of equipment that may impact storage or distribution of materials.
In addition to the items above, SIC agreements will define the method for reporting of changes. Typically, it will be an e-mail address to which change notifications and the supporting documentation can be forwarded. Some companies may also choose to have changes reported to a specific individual or department, either by electronic or hard-copy means. Some suppliers have customized mechanisms for ensuring that their customers are notified of changes. For example, a supplier may require that customers sign up for change notification announcements through an automated web form or similar tool. It is important that appropriate communication takes place between suppliers and customers to ensure that each other’s needs and systems are understood and met.