Why, Why, Why…ELISA? A Look at the Benchmark HCP Assay
March 14, 2019 - Cytiva
Real-Time Characterization of Biotherapeutics and Comparability o
March 14, 2019 - BioPharm International
Real-Time Characterization of Biotherapeutics and Comparability of Biosimilars
…
Bispecific Antibody Purification: Insights and Case Studies
February 28, 2019 - Cytiva
New modalities, like bispecific antibodies, present unique challenges compared to mAb production. For purification, there’s not just one right answer to which
…
Make the Switch from ELISA to Biacore™ SPR-Based Assays Today
February 28, 2019 - Cytiva
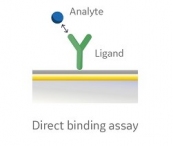 …
…
Protein Analysis with Size Exclusion Chromatography
February 28, 2019 - Cytiva
Size exclusion chromatography (SEC) is widely used in several different analytical applications from basic research to quality control of biotherapeutics. This
…
News: Cytiva Adds ELISA Kit to End-to-End HCP Monitoring Capabili
February 28, 2019 - Cytiva
Cytiva is pleased to announce the new Amers
…
Nurturing Knowledge from Disparate Data Streams
February 28, 2019 - BioPharm International
Reduce Hands-On Time in Your Ion Exchange Chromatography Runs
February 14, 2019 - Cytiva
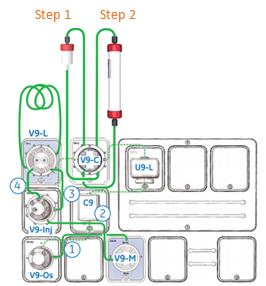 …
…
Single-Step, Protein A Chromatography Process for Bispecific Anti
February 14, 2019 - Cytiva
 A single-step downstream process using protein A chromatograp
A single-step downstream process using protein A chromatograp
Supply Chain Challenges for Single-Use Systems
February 14, 2019 - BioPharm International
Suppliers address the complexity of supplying disposable components to the global biopharmaceutical manufacturing industry.